Has MES Reached Maturity in Pharma Industry
Compliance, process optimisation, increasing profit, improving the supply chain and aligning to corporate standards are driving investment of Information Technology (IT) in the pharmaceutical industry. According to a report in October 2010 by Gartner Group, there is a need for integrated IT manufacturing technologies, reflected in the fact that corporate IT budgets have increased for manufacturing operations from 3% in 2001 to 19% in 2007 (1). The pharmaceutical industry has adopted this strategic initiative of integrating manufacturing IT and is benefiting through improved quality and production efficiencies.
Trend of IT in Manufacturing
IT can play a role at all levels of manufacturing operations however it is becoming especially evident within Manufacturing Operations Management (MOM) level. Advancements in technology such as Service Oriented Architecture (SOA) frameworks has enabled IT in manufacturing to shift from being data centric to process centric; therefore, IT is becoming seamless within manufacturing operations. By not restricting technology at the MOM level, activities such as Electronic Batch Records (EBR), real-time reporting, Enterprise Resource Planning (ERP) and equipment integration all have allowed pharmaceutical companies to maximise return on IT investment, improve quality, provide a platform for continuous improvement and ultimately increase profits.
MES in the Pharmaceuticals Industry
Manufacturing Execution Systems (MES) aggregates a number of the technologies deployed at the MOM level. MES as a technology has been successfully deployed within the pharmaceutical industry since the Food and Drug Administration (FDA) decreed the final 21 Part 11 regulations on 21 March 1997. These provided criteria for acceptance by the FDA, under certain circumstances, of electronic records, electronic signatures and handwritten signatures executed to electronic records as equivalent to paper records and handwritten signatures executed on paper. Over 16 years on and the technology associated with MES has matured positively and is fast becoming a recognised best practice across all life science regulated industries. This is borne out by the fact that green-field manufacturing sites are starting with an MES in place, that is, paperless manufacturing from day one.
The amount of IT applied to an MES project is dependent on the customer’s business needs. At a minimum, an MES should strive to replace paper batch records with an EBR. Other functionality that can be applied include automated material weigh & dispense and integration to ERP systems; therefore, helping the optimization of inventory levels and production planning. MES can also be integrated at the factory level, potentially giving complete control over the entire enterprise. This level of control helps ensure ‘right-first-time’ manufacturing and total enterprise visibility. The MES acts as a central system with effective interoperations with other manufacturing systems and departments such as operations, quality, maintenance and inventory control. The key to a successful MES implementation is applying the right level of IT to maximise Return on Investment (ROI).
This diagram is a representation of where MES would reside in a typical pharmaceutical facility in accordance with the ISA S95 standard (2). The ISA S95 standard defines a model for manufacturing operations, including the reporting and analysis functions that are critical to effective manufacturing. The business planning and logistics (Level 4) functions are supported by ERP, Product Lifecycle Management (PLM) or Supply Chain Management (SMC) applications. The plant floor systems (Levels 2 and 3) are made up of Laboratory Information Management Systems (LIMS), OPC data integration tools, data historians, Statistical Process Control (SPC), MES, control systems and database tools.
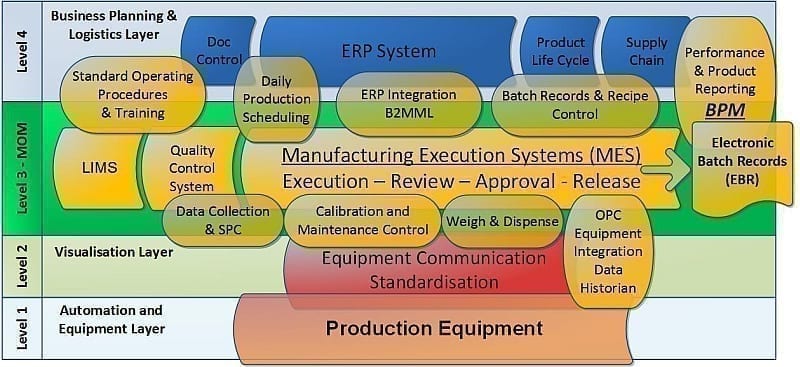
Figure 1.0: MES in Accordance with the ISA95 Model
Has the Pharmaceutical Industry Adopted MES?
In preparation for this article, a survey was carried out by the author to establish the level of electronic MES deployment within the pharmaceutical industry. The survey was conducted between January and February 2013. Over 400 senior managers and system analysts from across the globe working in pharmaceutical companies were contacted. In total, 84 responses were documented, of which 47% of respondents stated that electronic MES has already been deployed in specific manufacturing facilities. A further analysis was conducted of those facilities that had deployed MES in relation to stored batch records. Only 31% of companies had deployed full paperless solutions with the majority deploying a combination of paper and electronic batch records.
So what do the regulatory agencies think of MES? As far back as 2004, reports were published highlighting the compliance benefits of MES in the life science industry. In an article titled ‘MES Reduces FDA Compliance Costs’ (3), Quality Magazine discusses:
‘The underlying premise of today’s interpretation of the regulations is to ensure quality or risk management and risk mitigation by defining a management methodology for designing quality into the manufacturing process instead of attempting to build quality into products through inspection. The goal of manufacturers and the FDA is to provide and deliver safe and effective products.
However, FDA compliance historically has been an expensive, albeit necessary, proposition. As the life science industry becomes more competitive, reducing the cost of compliance is as important to success as controlling costs, quality and time-to-market.
A Manufacturing Execution System (MES) helps bridge the gap between regulatory control, operational control and case management.’
Since 2004, the FDA have audited pharmaceutical facilities with MES in place. It has been the experience of the author that these systems are welcomed by the FDA and there have been no citations against compliant MES implementations.
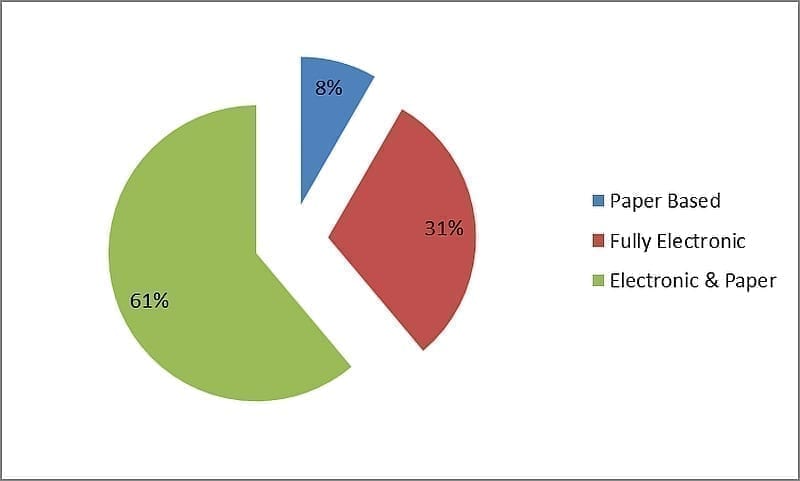
Figure 2.0: Batch Record Method with MES in the Pharmaceutical Industry
Examining the Principle Drivers for Adopting MES in the Pharmaceutical Industry
A GAMP® Guide to MES (4) states that the benefits for recipe-driven operations such as pharmaceutical production processes include:
- improved scheduling and resource utilization
- improved manufacturing flexibility and process changeover
- reduced Work in Progress (WIP) and improved material tracking
- shorter production cycles
- enforced sequence of operations
- reduced production record errors, electronic or hybrid
- improved visibility, accuracy and consistency of manufacturing data, enhancing decision support, Process Analytical Technology (PAT), and investigations capabilities
- minimized product recalls
- increased plant reliability
- realize paperless manufacturing
- automated Key Performance Indicator (KPI) generation and reporting, such as an Overall Equipment Efficiency (OEE) calculation
- support knowledge management and PAT
- reduce Quality Unit resources required for day to day operations by providing functionality, such as Electronic Production Records (EPR) and Review By Exception (RBE)’
As part of the survey conducted in preparation for this article, the respondents who had MES installed were asked to choose the primary and secondary drivers for implementing electronic MES solutions from the following five categories:
- Quality
- Production
- Inventory
- Financial
- Corporate
How does MES help the pharmaceutical industry?
The MES replaces paper-based processes that are error-prone and too slow for the modern-day pharmaceutical industry. It controls, monitors, and documents the processes digitally in real-time for the entire production cycle. Further, it boosts production efficiency and improves product quality, and could be easily integrated with your ERP.
The MES acts as a central system with effective communication flowing between manufacturing systems and other departments, including operations, quality, maintenance, and inventory control. Through real-time data that is available, it enables direct execution of production orders that can react dynamically to changing orders, machine statuses, quality checks and much more.
MES For Everyone to Gain a Competitive Edge
The MES solution contains intelligent software modules to help manage, analyse, and visualise production data quickly and easily, as per an organisation’s requirements. For instance, if a company is looking to reduce its Total Cost of Quality, an MES-based quality assurance module can be implemented to minimise significantly batch review times and lead times. They simultaneously increase the Right-First-Time (RFT) compared to a paper-based environment.
Pharmaceutical companies that have adopted MES are gaining a competitive advantage with improved quality, better traceability, maximised lean initiatives, flexible manufacturing processes, improved compliance, complete business process management, and accurate real-time reporting. Companies that do not adopt this technology risk losing a share of existing and new market opportunities.
The results of the survey can be seen here:
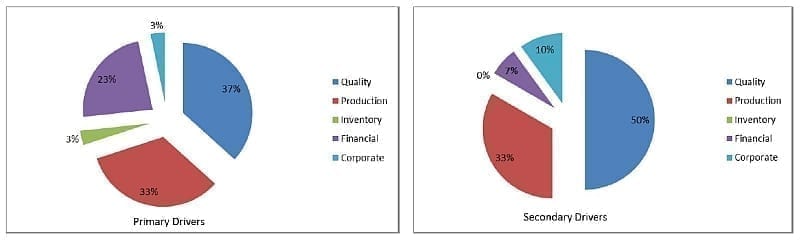
Figure 3.0: Primary and Secondary Drivers for MES in the Pharmaceutical Industry
From studying these results, it is evident that improving quality and production are the primary drivers for implementing electronic MES as they account for almost 80% of the combined drivers.
The benefits to MES can be quantitative and qualitative. Quantitative benefits can be measured for financial outcomes such as calculating an ROI, whereas qualitative benefits are more subjective, but no less important. When selling an MES internally, the importance of qualitative benefits should not be ignored, as inevitably measuring MES on quantitative benefits alone is a very hard sell.
Quantitative Benefits/Tangible Cost Saving to MES
Reduced Cost of Quality: The number of deviations associated with paper batch records is an example of the cost of quality. It is the direct experience of the author that with the implementation of MES, recorded deviations can be reduced by over 50%. With paper based systems, companies can maintain high levels of quality; however, there is a high cost associated with ensuring this quality level. An MES reduces the cost of quality but also ensures there is no drop in quality level.
Cost of Storing Batch Records (Quality): Companies have on-going costs associated with the storing and retrieval of paper batch records. With the introduction of an MES, major cost reductions can be achieved. (However, existing records still need to be held in accordance with regulations applied by the relevant agency.)
Improved Batch Release Time (Production): Review by exception can be achieved with a mature MES. EBRs typically require review and release by the operations and quality departments; however, this is significantly less than a paper review process. In some cases, pharmaceutical manufacturers have implemented Review-by-Exception with MES.
Production Capacity Increase: With an MES in place, companies can expect to improve the capacity of their manufacturing plant by maximising MES lean initiatives. This enables companies to take up any future or short-term demand without hiring extra resources or overtime.
Better Financial Costing: MES can help enable companies implement multi-level Bill of Materials (BOMs) with increased visibility on the manufacturing. Also, MES will lead to improved routing of material and better analysis of variance. It will enable the setting up new cost centres and provide all-in-all improved standard costing through the manufacturing process.
Inventory Reductions: Companies with little visibility into the manufacturing floor maintain a high level of inventory. MES solutions create near real-time reports using quality-approved data therefore allowing companies significant scope to reduce inventory levels.
Inventory Management: An MES improves warehouse efficiencies through stock movements and transactions being real time, paperless cycle counting and benefits associated with a paperless warehouse. This means there is a reduction in warehouse activities and a reduced effort for the quality department of incoming inspection labelling.
Qualitative Benefits/Intangible Cost Savings to MES
Enforced Compliance (Quality): MES provides enforced compliance in many aspects of manufacturing including enforced sequence of activities, equipment usability verification prior to use, material status checking prior to use, user group membership prior to performing system functions and many more.
Cost of Audit Preparation (Quality): MES helps close open deviations by putting in place rigid corrective actions such as enforced in-process inspections. This compares to a paper system that will rely on procedural updates and training, which are not as effective as an enforced quality check. During audits, MES is more efficient at retrieving information, which means shorter and better audits.
Electronic Equipment Management (Production): MES enables the efficient creation and automatic maintenance of electronic logbooks. Comprehensive status monitoring effectively prevents the use of wrong equipment. These equipment states might include planned or unplanned maintenance, cleaning and calibration. Business rules can be applied to manage the status of the equipment. MES can ensure that work in progress is not processed through a particular machine when flagged for being out of compliance.
Multiple Batch/Shift Production: One of the biggest benefits from MES is product changeover and new product Master Batch Record (MBR) creation or updates. The more products handled in production or packaging, the less effective paper-based systems become.
Transparent Process Data Evaluation (Corporate): MES stores data in a relational format that is available for integration and analysis with other Opcenter Intelligence information.
Business Process Standardization (Corporate): An MES program can be used as a mechanism to define and implement common business processes as well as lean manufacturing processes.
Untapped Capital (Financial): MES accelerates the manufacturing learning curve across all facets of the organisation because an MES implementation requires complete involvement and transparency of roles between different departments. This interaction between departments will unleash actionable intelligence, that is, employees adding valuable input into other functional areas. Employees can be empowered with new more value-added roles now that the control of the manufacturing process is automated.
Support the Extended Supply Chain Strategy (Inventory): MES solutions typically integrate with ERP systems. MES helps refine ERP scheduling functions to the day or even hour, whereas the ERP focuses on months or weeks. This extra level of planning helps ensure that order times are minimised and there is increased visibility into the manufacturing floor.
Corporate Business Process Management (BPM): BPM is the activity of managing your whole process, from dock to stock as well as supporting functions. As part of a demand-driven manufacturing process, increasing efficiency is critical. Real-time information from the manufacturing process is needed to prevent problems, drive quality, and enable the flexibility your customers require. MES enables this reach into the manufacturing process and is seen as a key requirement for successful BPM.
Challenges Associated with an MES Implementation
An MES project within the pharmaceutical industry is treated as an enterprise software solution, similar to ERP projects. However, the major difference between MES and ERP projects is that MES should be led by operations whereas ERP projects are typically IT led. MES touches every aspect of the manufacturing floor and can become very complex. For this reason, companies have concentrated on ERP and automation projects as they do not have as many dependencies. MES is often the final hurdle to complete data integration across the ISA S95 model.
Implementing a complete MES solution into an existing pharmaceutical manufacturing facility could take in excess of four years. In year one, the strategy applied could be to expand the functionality of the ERP layer with improved material tracking on the production floor and warehouse. Lean initiatives can be implemented to ensure that the manufacturing and business processes are ‘MES ready’.
Into year two, focus might shift to investing into the visualisation layer of automation, such as investments into Distributed Control Systems (DCS) and Data Historian technologies. The strategy might also involve investing into an automatic weight & dispense solution, either provided by an ERP or MES vendor; therefore, improving the control of raw materials on the production floor. On to year three and the company is now ‘MES ready’. An MES solution can be deployed with the purpose of replacing a paper batch record with an EBR. Finally in year four, the focus will shift to fully integrating the MES solution into the automation and ERP layers.
MES teams typically consist of a steering committee or group with sub-groups concentrating on production, quality, validation, IT, change management, inventory, training and maintenance requirements. Group sizes can be from 6 to over 20 directly involved in the MES project. Factoring in the human resources required, time scale, software licenses, hardware costs, initial negative impact of change on production, a budget of 3-5 million euros is not unusual for an MES project deployed in a small to medium sized pharmaceutical operation, that is 200-500 employees.
MES solutions are being successfully deployed in the pharmaceutical industry; however, deciding to implement an MES solution is not a decision to be taken lightly. Careful planning and education into what MES is, is key. Organisations such as the Manufacturing Enterprise Solutions Association (MESA) (6) have been established to promote best practices and education in MES. With proper planning and education, a business case for an MES solution can be established.
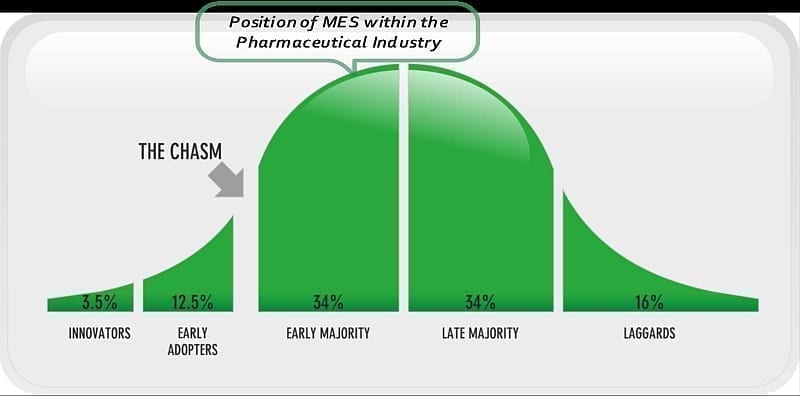
Figure 4.0: Crossing the Chasm of MES in the Pharmaceutical Industry
The Technology Adoption Cycle and MES in the Pharmaceutical Industry
Geoffrey A. Moore in this book ‘Crossing the Chasm’ (5) discusses a concept of technology adoption. Moore breaks technology adoption into five phases. Cracks exist between the phases and some technologies that fail to clear these cracks fade away. Examples of such technologies that were not fully adopted or did not reach maturity include Windows ME, the PalmPilot and social networking site Bebo. Moore (5) goes onto discuss ‘the notion that part of what defines a high-tech market is the tendency of its members to reference each other when making buying decisions is absolutely key to successful high-tech marketing.’ The critical point identified by Moore where technologies either get adopted or fail is the chasm, the crack that exists between the ‘Early Adopters’ and ‘Early Majority’ phases.
So has MES in the pharmaceutical industry crossed the chasm? Based on the research conducted in conjunction with this article, yes it has. It is firmly placed in the ‘Early Majority’ phase as demonstrated in the following diagram.
People who are deemed to be in the ‘Early Majority’ phase are pragmatists. Moore (5) says about the Early Majority that ‘they care about the company they are buying from, the quality of the product they are buying, the infrastructure of supporting products and system interfaces, and the reliability of the service they are going to get…’ All these factors are very important considerations when selecting a technology in the pharmaceutical industry. There are thousands of MES products available. MES is being deployed in the pharmaceutical industry and there is an array of proven solutions available.
Our Digital Rising is a strategic procedural method that fully enables manufacturing businesses to realise their digital ambitions irrespective of size and scope. It is a holistic approach combining traditional, modern, and innovative technologies with in-depth knowledge of integration and communications. The primary focus of Digital Rising is to identify the challenges faced, understand short-term requirements, and chalk out an innovative and futuristic long-term objective.
It is a confluence of MES, Pharma 4.0, AI, and Industrial Internet of Things (IIoT) that can accelerate the digitalisation journey of the pharma industry.
The S-Curve and ERP in the Pharmaceutical Industry
Another methodology used to describe technology adoption is the S-Curve. The S-Curve describes technological evolution and suggests that technologies evolve through an initial period of slow growth, followed by one of fast growth culminating in a plateau. It also breaks the take up of technologies into different phases; however, it differs from Moore’s (5) model in that it accounts for shifts in technology during the adoption cycle. Traditionally, MES has been a site-specific initiative but with increasing globalisation, companies are now aligning not just their enterprise systems such as ERP but are focusing on manufacturing systems such as MES. Software as a Service (SaaS) and cloud computing is arguably causing a shift in the technology adoption cycle in both MES and ERP. Cloud computing is a model whereby the computer hardware and software services are typically (but not always) offered as a service in a remote data center. This compares to deploying hardware and software on local sites.
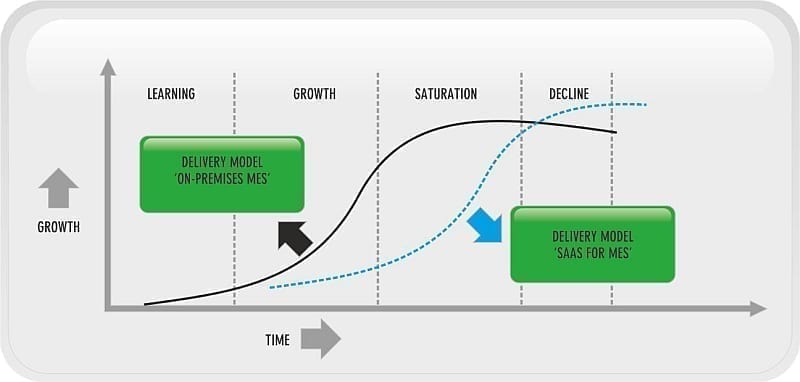
Figure 5.0: S-Curve Technology Adoption
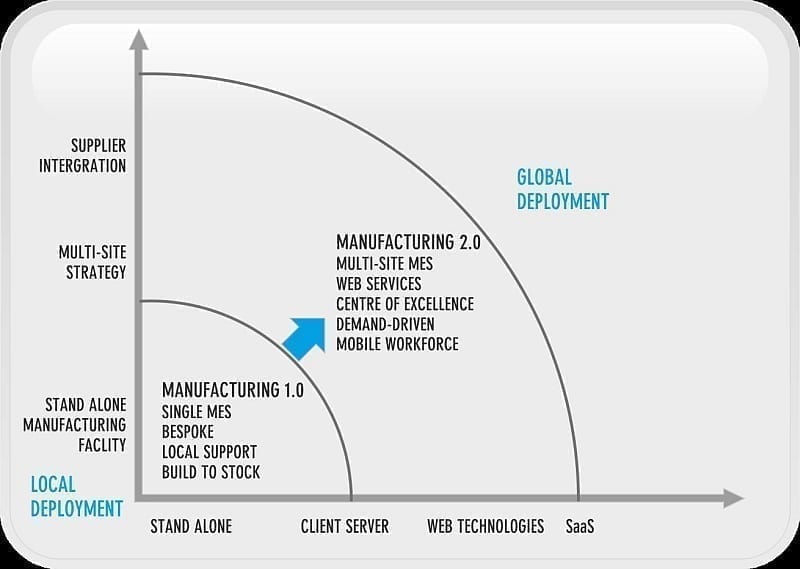
Figure 6.0: Manufacturing 2.0 and MES
Manufacturing 2.0
With emerging technologies such as cloud computing and increasing globalisation, the dynamics of manufacturing are changing. Manufacturing plants are integral parts of the overall supply chain and need to provide real-time data. This increases the focus on reporting from manufacturing and MES plays a crucial role in insuring the accuracy and availability of data. ERP systems handle weekly or perhaps in some cases daily information. The supply chain is demanding more refined data. MES solutions typically are designed to report at an hourly rate or with less frequency, therefore meeting the requirement of the overarching supply chain.
MESA, in a recent white paper, introduces the concept of Manufacturing 2.0 (Mfg 2.0). As discussed earlier, companies are shifting manufacturing focus from data centric to process centric, and advances in the design of large software applications, through the use of frameworks such as Service Oriented Architecture (SOA) are enabling this shift. MESA discuss (7):
‘The manufacturing operations‐specific requirements for SOA are called Manufacturing 2.0. Mfg 2.0 is differentiated from the so‐called Manufacturing 1.0 architectures based on stand‐alone client/server data base applications that attempted to represent business process modelling through point‐to point interfaces and custom data transformation between applications.’
By deploying Mfg 2.0 technologies, companies can meet the demands driven by the overall supply chain. Without an MES, this cannot be achieved efficiently. Companies will have to decide whether Mfg 2.0 technologies are feasible for the deployment of MES. The technology is not as mature as client/server based solutions; concerns exist in relation to data security and a total reliance of IT networks. Individual companies should weigh up benefits of these new technologies, such as economies of scale, shared services and aligning to enterprise strategies, before deciding on a deployment approach of a technology set.
Pharmaceutical Manufacturing Without an Electronic MES
Due to the complexities at the MOM level, financial constraints and availability of key personnel investments in solutions such as MES are deferred. This is very understandable as manufacturing organisations need to be ready and able to clearly define their requirements for a potential solution. The downside to this strategy is a break in data flow, that is, inventory information available on the ERP system and process information available from equipment sets, but no one system aggregating the data together. This impacts on the availability and quality of data, therefore negatively impacting on traceability. It is also a requirement within FDA-regulated industry to retain MOM data associated with products and processes that directly impact on the production of finished goods. With e-pedigree and serialisation pending, this break in data flow requires companies to invest a lot of time in managing the data on paper.
This data is critical in the pharmaceutical industry. It is a rich, real-time and most importantly a quality approved (that is, collected, stored and maintained in accordance to regulatory requirements) source of data on which any decisions pertaining to manufacturing can confidently be made. The data will also help resolve traceability issues arising at the MOM level, as it plugs the gap between enterprise and manufacturing operations management.
In Conclusion
Pharmaceutical companies that have adopted MES are gaining a competitive advantage with improved quality, better traceability, maximised lean initiatives, flexible manufacturing processes, improved compliance, complete business process management and accurate, real-time reporting. Companies that do not adopt this technology risk losing a share of existing and new market opportunities. This is borne out by the fact that new pharmaceutical facilities are starting with an MES in place, that is, paperless manufacturing from day one.
Of those companies surveyed in conjunction with the article, over 80% have an ERP system in place. MES adoption is lagging behind ERP, but as Mfg 2.0 becomes more prevalent, MES will become an integral part and critical component of the overall supply chain. MES will continue to be adopted and eventually align with ERP.
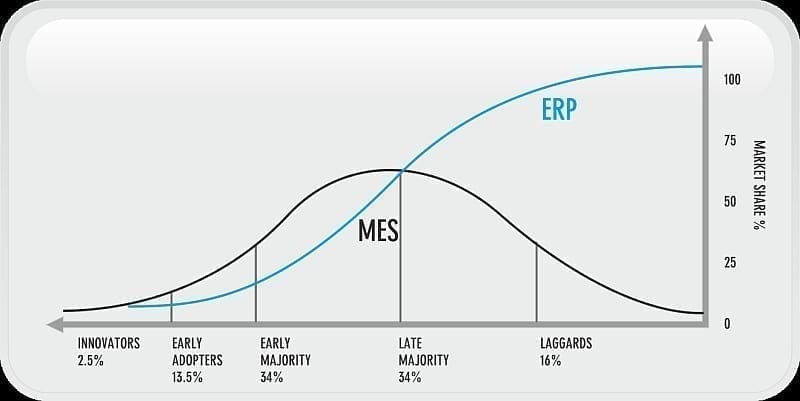
Figure 7.0: MES and ERP Adoption in the Pharmaceutical Industry
Implementing MES is not a decision that can be taken lightly. Every aspect of the manufacturing process will be impacted by an MES implementation; however, companies are successfully deploying MES and reaping the reward. MES has crossed the technology chasm and is reaching maturity in the pharmaceutical industry. Don’t get left behind.


